Breast augmentation has come a long way in terms of safety and reliability, and continues to be one of the top cosmetic surgical procedures in the world. The industry is expected to grow even more, with groundbreaking technology such as Motiva breast implants, the 6th generation of breast implant technology that is a trailblazer in safety and innovation.
Motiva Ergonomix® Implants Singapore

Having breast augmentation surgery is a lifetime commitment, hence finding the right implants from the company that puts your safety above everything else is Vital.
To meet the demands of the market regarding high-quality implants that have the look and feel of natural breasts, while at the same time prioritizing safety and reliability, Motiva launched the Motiva Ergonomix® implants, the 6th generation of breast implants.
From the word “Ergonomix,” Motiva breast implants were designed to mimic the real shape, feel and movement of natural breasts, giving emphasis on comfort and durability, taking into account the active lifestyle of women in this century.
Motiva Ergonomix® moves as you move. The implants are designed to take the shape of natural breasts, conforming to your movement whether standing up or lying down.
Motiva Implants Singapore: Safety through Innovation
The shape and feel of implants is one thing, while safety is another. Motiva made it a point to address the shortcomings of previous implant designs and technology in the industry, creating the 6th generation implant that leads the industry in safety and innovation.
ProgressiveGel Ultima™
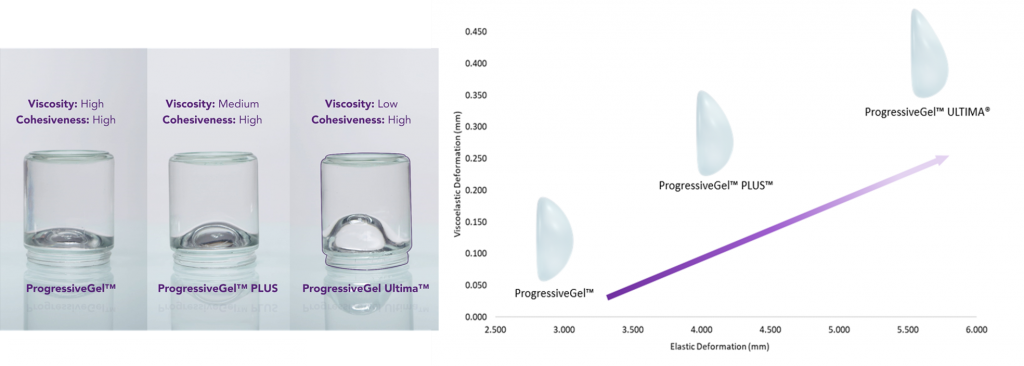
Motiva Ergonomix® makes use of “ProgressiveGel Ultima™,” a patented silicone gel that does not only provide the soft feel of natural breasts but also prevents gel-bleeding in case of rupture.
The ProgressiveGel Ultima™ was also designed to allow for faster recovery and easier insertion, making the significantly safer and your healing process much faster.
Q Inside Safety Technology™

Another feature of the implant is the “Q Inside Safety Technology™,” that allows you to verify all the characteristics of your breast implants, including product family, volume, serial number and lot number.
The Q Inside Safety Technology™ makes use of the QID Reader, a device that reads the electronic serial number embedded in your implant’s micro transponder, which allows you to authenticate your implants in a non-invasive way.
You can also access the “MotivaImagine App” for updates on written policies and procedures regarding your product warranty and the possibility of financial support in case of a replacement surgery.
BluSeal

As a patented visual safety indicator, all Motiva implants appear slightly blue when intact. The color signifies to surgeons that the entire implant shell barrier layer is intact before insertion.
TrueMonobloc®

All Motiva implants are made as a flexible, multilayer system that links all implant components (gel, shell, and patch) to create a unified structure that allows for easier insertion, robust function, and a clinically proven safety profile.
SmoothSilk™/SilkSurface™

All Motiva implants are housed in the proprietary “SmoothSilk™/SilkSurface™” shell with smooth, 360-degree uniform NanoSurface™ technology, that has been proven to minimize inflammation and inhibits the formation of biofilm or bacteria around the implant.
The SmoothSilk™/SilkSurface™ shell also reduces the risk of double capsules and late seromas and enable implant insertion via smaller incisions.
Motiva Implants Singapore: Tried and Tested
To prove the safety and reliability of its products, Motiva took the initiative to conduct a clinical study with 233 patients to test for any presence of complications, such as implant rupture, capsular contracture, double capsules or late seromas.
This evaluation was monocentric, conducted at a Plastic Surgery Center accredited by the American Association for Accreditation of Surgery Facilities, by a group of board certified Plastic and Reconstructive
Surgeons.
After the 6th year follow-up, there have been no reported rupture, no capsular contracture, no double capsule or late seromas and no implant replacement due to clinical reasons.
Motiva has placed a total of 302,124 breast implants in the international market since its commercial launch in 2010, and since then has only had 0.025% clinical related events reported, with no cases of implant rupture due to device failure.
The patients involved in the study also reported an aesthetic satisfaction rate for satisfied to extremely satisfied, with majority of them belonging to the extremely satisfied group. This proves that Motiva Implants® are not only safe, but also fulfills their aesthetic preference.
Motiva Implants Singapore: Long-Term Security
Motiva values the people that use their products, and provides them the security that they deserve by giving free replacement in the event of rupture for the lifetime of the implant. The company also provides assistance in cases of capsular contracture Baker Grades III and IV through its product replacement policy program.
There are also 2 extended warranty programs in the event of a rupture or capsular contracture Baker Grades III or IV, that include financial assistance application to the revision surgery in the event of implant replacement.
Extended Warranty Program:
- 5 years Motiva Program™ – $2,500 financial assistance per affected implant for Motiva Implants® with Q Inside Safety Technology™.
- 2 years Motiva Program™ – $1,000 financial assistance per affected implant for Motiva Implants® with Q Inside Safety Technology™.





